
The results are in and we are very proud to share the results of our first major FDA study for OsNovum!
Our study for FDA clearance started in 2016 and we have now received the results. This was a highly controlled study that requires registration with the FDA and the USDA. Over a thousand pages of documents have been compiled to comply with regulatory requirements. These studies also required us to hire independent, 3rd party quality control personnel to monitor all phases of the study. The FDA reviews all documents and spends a full week inspecting our processes. Additionally, OsNovum hired an outside laboratory to perform the histopathological analysis of our study and we are now reporting on their findings.
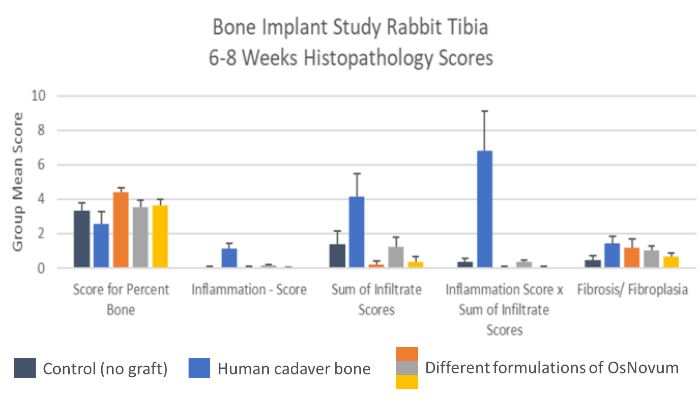
The primary objective of the study is to evaluate the inflammatory response to our material to make sure it is safe for implantation into humans. The second aspect is to confirm that the material is effective for its intended purpose, which in our case is to grow bone. OsNovum has outperformed in both aspects. We are required to compare our material to a known graft material and in this case, we chose the most commonly used material: human cadaver bone.
We also compared OsNovum to no graft (control). We choose three formulations of OsNovum to be evaluated in the test. OsNovum produced more bone than the commonly used cadaver bone and more bone than the control. These findings prove that OsNovum stimulates bone growth, but more importantly for the FDA, OsNovum produced less inflammation than no graft and many times less inflammation than cadaver bone. While this is the most significant test for FDA clearance, we also expect similar findings for the additional tests that your investment is funding.
This is a major step forward for OsNovum!
"OsNovum Nearing $150K in Funding"
"New Methods of Diagnosing Who Will Suffer a Bone Fracture"
Tissue Engineering and Regenerative Medicine International Society (TERMIS)
American Society for Bone and Mineral Research (ASBMR)